Finally, the data is in here at OZMOSI, and we can start tracking and reporting on the clinical development expected to come out of China. Gone are the days of developing drugs only in the US and then watching them slowly make their way to the rest of the world.
China’s current trial pipeline below illustrates a disease area focus that is on par with what we see and expect from the US, EU, and Japan.
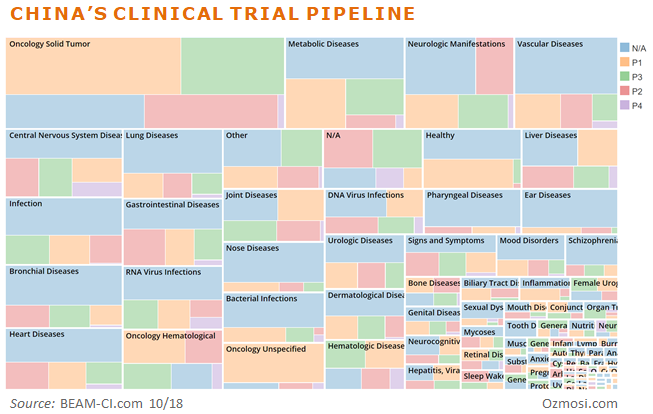
And that’s only the beginning. China is now taking a very active role in developing novel therapies directly, and not just for their own country. We are starting to see drugs developed in China first and then get picked up by companies in other countries.
Looking at one of the hottest treatment approaches today, CAR-T cell therapy accounts for 5% of China’s current ongoing trials across almost 30 different diseases.
Don’t just look for the novel items either, we are expecting to see quality biosimilars showing up in western markets from China as well.
If you don’t have a careful watch on China’s clinical development pipeline, you are in for some unpleasant surprises in the near future.
